 When one mole each of gaseous Na + and Cl – ions form solid NaCl, 769 kJ of heat is released. Lattice energy is the amount of energy released when. Thus, it requires 769 kJ to separate one mole of solid NaCl into gaseous Na + and Cl – ions. This chemistry video tutorial provides a basic introduction into the lattice energy of ionic compounds. 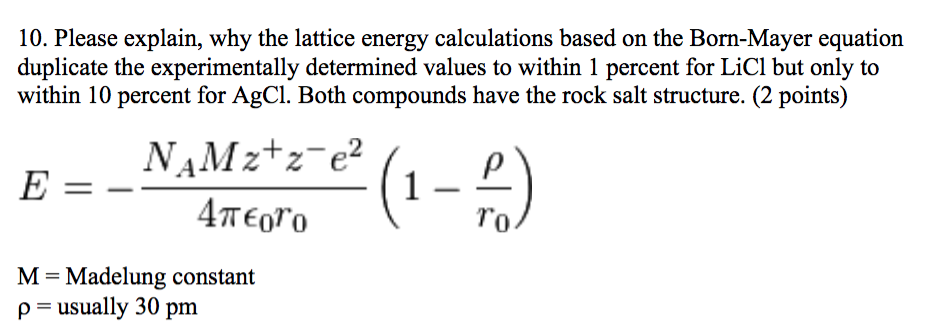
The medium or solvent has a dielectric constant. In the discussion of lattice energy, we consider the ions separated into a gas form whereas in the dissolution process, the ions are also separated, but this time into ions dispersed in a medium with solvent molecules between ions. For sodium chloride, Δ H lattice = 769 kJ. Relating Hydration Energy to Lattice Enthalpy. In both cases, a larger magnitude for lattice energy indicates a more stable ionic compound. Thus, if you are looking up lattice energies in another reference, be certain to check which definition is being used. Some texts use the equivalent but opposite convention, defining lattice energy as the energy released when separate ions combine to form a lattice and giving negative (exothermic) values. However, the lattice energy can be calculated using the equation given in the previous section or by using a thermochemical cycle. In the Kapustinskii formula, the lattice energy (kJ/mol) is given by: (5.8.3.1) E L 1213.8 z + z n r + + r ( 1 0.345 r + + r ) Here the sum of the monovalent radii is used in place of r o, the bond distance in the Born.  
Note that we are using the convention where the ionic solid is separated into ions, so our lattice energies will be endothermic (positive values). He thus arrived at a lattice energy formula using an average Madelung constant, corrected to monovalent radii. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
